Toward Pharmaceutical Sovereignty in Africa: Regulatory Transformation, Industrial Development, and Regional Integration
DOI:
https://doi.org/10.51412/psnnjp.2026.001Keywords:
pharmaceutical sovereignty, local pharmaceutical manufacturing, regulatory governance, AfCFTA, medicine security, African pharmaceutical industryAbstract
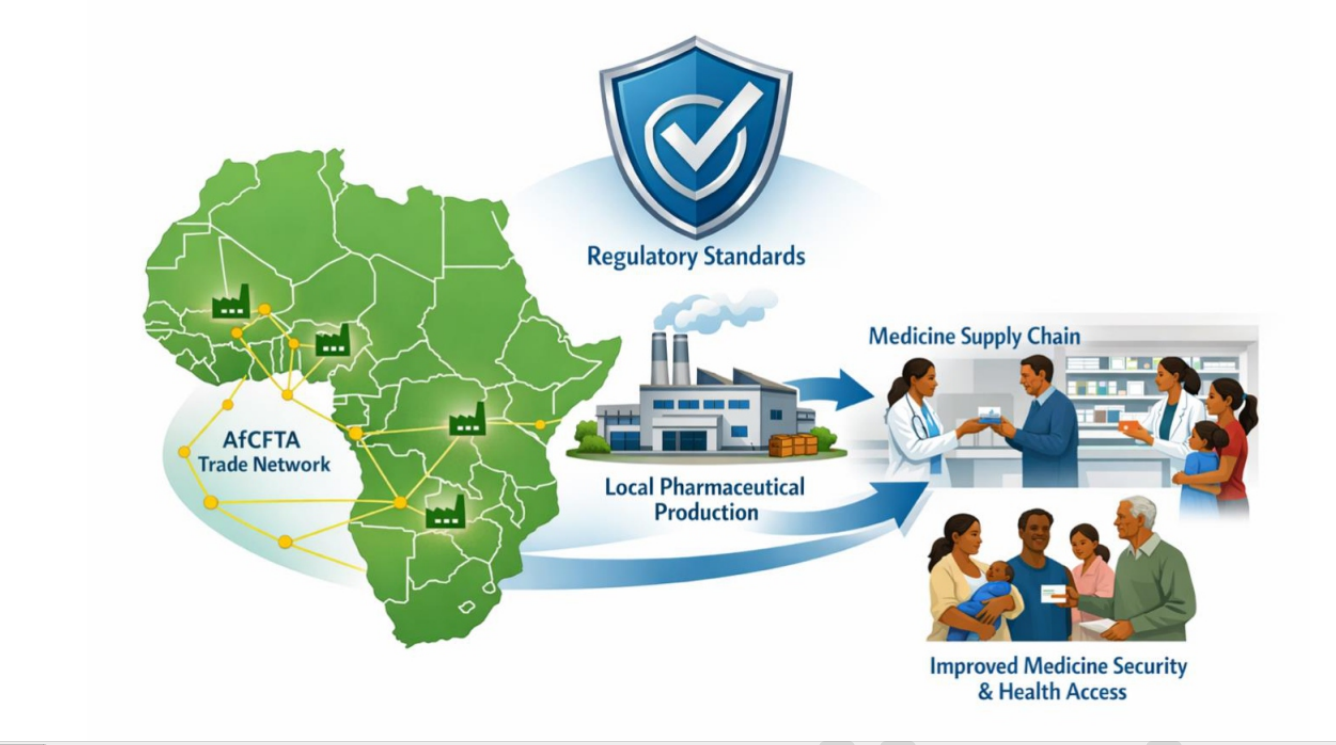
Africa's pharmaceutical sector is undergoing a significant structural transition as governments, regulators, and domestic manufacturers seek to reduce reliance on imported medicines and strengthen local production capacity. Historically, pharmaceutical markets across the continent were largely shaped by multinational manufacturers, while indigenous firms faced constraints due to regulatory uncertainty, limited access to capital, and fragmented industrial policy frameworks. Recent market changes—including the partial withdrawal or restructuring of multinational operations in several African countries between 2022 and 2024—have created both supply concerns and strategic opportunities for domestic industry expansion. This study analyses the evolving pharmaceutical landscape across Africa, with Nigeria serving as an illustrative case. Drawing on policy analysis, market trend evaluation, and literature synthesis covering the period 2024–2026, the paper examines how regulatory strengthening, industrial investment, and regional trade integration are influencing the continent's pharmaceutical trajectory. Particular attention is given to the evolving roles of regulatory institutions such as the National Agency for Food and Drug Administration and Control (NAFDAC) and the Pharmacists Council of Nigeria (PCN) in reinforcing quality assurance systems and supporting domestic manufacturing growth. The analysis suggests that credible regulatory governance, coordinated industrial policy, and expanded regional trade through the African Continental Free Trade Area (AfCFTA) may enable Africa to transition toward pharmaceutical sovereignty gradually. If sustained, these developments could foster the emergence of regionally competitive pharmaceutical manufacturers while strengthening medicine security and health system resilience.References
1. World Health Organisation (2023) Local Production and Access to Medicines
2. UNCTAD (2025). Building the Case for Investment in Local Pharmaceutical Production in Africa.
3. World Health Organisation (2025) World Local Production Forum.
4. Grand View Research (2024). Africa Pharmaceutical Market Size and Trends Analysis Report.
5. Credence Research (2025) . Africa Pharmaceutical Market Growth Forecast.
6. Institute for Health Metrics and Evaluation (2023). Global Health Data Exchange
7. African Development Bank (2024 ). Industrialisation and Health Systems Report.
8. AfCFTA Secretariat (2021). Agreement Establishing the African Continental Free Trade Area.Accra.
9. Persistence Market Research (2025) Africa Pharmaceuticals Market Forecast 2025–2032.
10. World Health Organisation (2022). Global Benchmarking Tool for Evaluation of National Regulatory Systems.
11. IQVIA , 2024 ; IQVIA , 2024 . https://www.iqvia.com / - /media/iqvia/pdfs/china/viewpoints/iqviainstitute-general-use-of-medicines-2024-forprint.pdf
12. Frost & Sullivan, 2024
13. Deloitte , 2024 Deloitte , 2 0 2 4 , [https://www.deloitte.com/ng/en/services/auditassurance/analysis/Audit-TransparencyReport.html](https://www.deloitte.com/ng/en/services/audit%02assurance/analysis/Audit-Transparency%02Report.html)
14. World Bank (2024) World Development Indicators.
15. ICH (2024) Int e rnational Counc il for Harmonisation Membership and Regulatory Harmonisation.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Prof. Lere Baale, FPSN, FPCPharm, FNAPharm,

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.


