The Role of l-arginine in prevention of testicular function toxicity induced by monosodium glutamate burden in Wistar rats
https://doi.org/10.51412/psnnjp.2022.25
Keywords:
Sperm morphology, Sperm motility, Semen quality, Testes histomorphology, Testosterone activityAbstract
Background: Monosodium glutamate, MSG, is a widely consumed and inadvertently abused glutamate-based flavour enhancer that could impair male fertility. L-arginine, is a fatherhood amino acid that could improve male reproductive apparatus. Possibility of simultaneous consumption of MSG and l-arginine exists with unknown outcomes on testicular function. This study aimed to evaluate the role of l-arginine in prevention of testicular function toxicity induced by monosodium glutamate burden in Wistar rats.
Methods: The study involved five rat groups (n = 5). Group 1 rats, normal control, received distilled water. Groups 2,3,4 and 5 rats, respectively received MSG (8000 mg/kg), l-arginine (60 mg/kg), MSG (8000 mg/kg) plus l-arginine (60 mg/kg) and MSG (8000 mg/kg) plus l-arginine (120 mg/kg). Exposure was by oral intubation for 28 consecutive days. Testicular function indicators studied included testosterone activity (in the rats' testes homogenate and serum), sperm quality (sperm count, sperm motility, sperm volume and sperm morphology) and testes histomorphology.
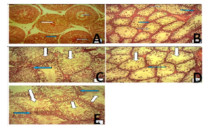
Results: Results revealed significant (P<0.05) and marked diminution of testosterone activity (in the testes and serum), sperm quality and spermatogenic cells of the testes in the rats that received MSG compared to rats in normal control and other groups, notably group 5 that received MSG and high dose of l-arginine together.
Conclusion: The study demonstrated that MSG burden caused testicular function toxicity in the rats, demonstrated by the altered testes histomorphology, diminished testosterone activity and compromised sperm quality. There was a significant reversal by l-arginine notably at 120 mg/kg of the testicular function toxicity caused by MSG burden in the rats via probable modulation of the compromised indicators as determined in the rats. Thus, l-arginine could play a significant role in the prevention of testicular function toxicity induced by MSG burden in rats.
References
Kayode OT, Rotimi DE, Kayode AAA, Olaolu TD, and Adeyemi OS (2020). Monosodium glutamate (MSG)-induced male reproductive dysfunction: A mini review. Toxics, 8(1): 7. https://doi.org/10.3390/toxics8010007
Fatin FJ, Ramya DM, Mahanem MN, Izatus ST and Siti BB (2019) Monosodium glutamate daily oral supplementation: study of its effects on male reproductive system on rat model. Systems Biology in Reproductive Medicine, 65(3): 194-204. https://doi.org/10.1080/19396368.2019.1573274
Jubaidi JF, Mathialagan RD, Noor MM, Taib I and Budin SB (2019). Monosodium glutamate daily oral supplementation: Study of its effects on male reproductive system on rat model. Systems Biology in Reproductive Medicine, 65(3): 194-204.
https://doi.org/10.1080/19396368.2019.1573274
Igwebuike U, Ochiogu I, Ihedinihu B, Ikokide J and Idika I (2011). The effect of oral administration of monosodium glutamate (MSG) on the testicular morphology and caudaepididymal sperm reserves of young and adult male rats. Veterinarski Arhiv, 81: 525-534.
Egbuonu ACC, Ejikeme PM and Obasi NL (2010). Monosodium glutamate: Potentials at inducing prostate pathologies in male Wistar rats. African Journal of Biotechnology, 9(36): 5950-5954. https://doi.org/10.5897/AJB10.635
Ezeanyika LUS and Egbuonu ACC (2011). Impact of nitric oxide and insulin resistance on the pathophysiology of the metabolic syndrome: Possible role of l-arginine and glutamate. Journal of Medicine and Medical Sciences, 2(2): 657-662.
Szlas A, Kurek JM and Krejpcio Z (2022). The potential of l-Arginine in prevention and treatment of disturbed carbohydrate and lipid metabolism - A review. Nutrients, 14: 961. https://doi.org/10.3390/nu14050961
Kumar N and Singh AK (2015). Trends of male factor infertility, an important cause of infertility: A review of literature. Journal of Human Reproductive Science, 8: 191-196.
Owoeye O, Oluwagbemi AD and Thomas MA(2018). Telfairia occidentalis leaf extract mitigated monosodium glutamate-induced behavioural and histological alterations in rat hippocampus. African Journal of Medicine and Medical Sciences, 47: 405-413.
Egbuonu ACC, Opara CI, Akachukwu D and Onyedikachi UB (2018). Effect of ethanolic extract of avocado pear (Persea americana) seed on normal and monosodium glutamatecompromised rats' hepatic histo-morphology and serum bio-functional parameters. Research Journal of Environmental Sciences, 12(2):53-62. https://doi.org/10.3923/rjes.2018.53.62
Egbuonu ACC, Obidoa O, Ezeokonkwo CA, Ejikeme PM and Ezeanyika LUS. (2010). Some biochemi c a l effe c ts of sub- a cut e or a l administration of l-arginine on monosodium glutamate-fed Wistar albino rats 1: Body weight change, serum cholesterol, creatinine and sodium ion concentrations. Toxicological and Environmental Chemistry, 92(7): 1331-1337. https://doi.org/10.1080/02772240903450645
NRC (National Research Council, USA) (2011). Guide for the care and use of laboratory animals. 8th Edition, Committee for the Update of the Guide for the Care and Use of Laboratory Animals, National Research Council, National Academies Press, Washington DC, USA.
Handelsman DJ, Jimenez M, Singh GKS, Spaliviero J, Desai R and Walters KA (2015). Measurement of testosterone by immunoassays and mass spectrometry in mouse serum, testicular, and ovarian extracts. Endocrinology, 156(1): 400-405. https://doi.org/10.1210/en.2014-1664
Maree L and Vande r-Horst G (2013). Quantification and identification of sperm subpopulation using computer-aided sperm analysis and species specific cut-off values for swimming speed. Biotechnic & Histochemistry, (3-4):181-193.
Vander-Horst G and Maree L. (2009). SpermBlue: A new universal strain for human and animal sperm which is also amenable to automated sperm morphology analysis. Biotechnic & Histochemistry, 84(6): 299-308.
Badkoobeh P, Parivar K, Kalantar SM, Hosseini SD and Slabar A(2013). Effect of nano-zinc oxide on doxorubicin-induced oxidative stress and sperm disorders in adult male Wistar rats. Iranian Journal of Reproductive Medicine, 11(9): 355-364.
Yeung CH, Anapolski M and Cooper TG (2002). Measurement of volume changes in mouse spermatozoa using an electronic sizing analyzer and a flow cytometer validation and application to an infertile mouse model. Journal of Andrology, 23: 522-528.
Ibrahim KE, Al-Mutary MG, Bakhiet AO and Khan HA (2018). Histopathology of the liver, kidney, and spleen of mice exposed to gold nanoparticles. Molecules; 23(1848): 1-14.
Oduwole OO, Peltoketo H and Huhtaniemi IT(2018). Role of follicle-stimulating hormone in spermatogenesis. Frontiers in Endocrinology, 9: 763. https://doi.org/10.3389/fendo.2018.00763
Kianifard D, Ehsani A, Daneshgar PZ, Akbari G and Shoar SMM (2019). Effect of monosodium glutamate on testicular tissue of paclitaxel-treated mice: An experimental study. International Journal of Reproductive Biomedicine, 17(11): 819 - 830.
https://doi.org/10.18502/ijrm.v17i10.5492
Xiao J, Zhaojian L, Xin R, Pengyuan D, Yansen L and Chunmei L (2020). L-arginine alleviates the testosterone reduction in heat-treated mice by upregulating LH secretion, the testicular antioxidant syst em and expr e ssion of steroidogenesis-related genes. Reproduction, Fertility and Development, 32(10): 885-892. https://doi.org/10.1071/RD19303
Estumano DP, Ferreira LO, Bezerra PAL, da Silva MCP, Jardim GC, Santos GFS, Gustavo KS, Mattos BG, Ramos JAB, Jóia de Mello V, da Costa ET, Lopes DCF and Hamoy M (2019). Alteration of testosterone levels changes brain wave activity patterns and induces aggressive behavior in rats. Frontiers in Endocrinology, 10: 654. https://doi.org/10.3389/fendo.2019.0065
Alalwani AD (2014). Monosodium glutamate induced testicular lesions in rats (histological study). Middle East Fertility Society Journal, 19(4): 274 - 280. https://doi.org/10.1016/j.mefs.2013.09.003
Mondal M, Sarkar K, Nath PPand Paul G (2018). Monosodium glutamate suppresses the female reproductive function by impairing the functions of ovary and uterus in rat. Environmental Toxicology, 33(2): 198-208.
Santi D, Crépieux P, Reiter E, Spaggiari G, Brigante G, Casarini L, Rochira V and Simoni M (2020). Follicle-stimulating hormone (FSH) action on spermatogenesis: A focus on physiological and therapeutic roles. Journal of Clinical Medicine, 9(1014): 1-27.
Akhigbe R and Ajayi A (2020). Testicular toxicity following chronic codeine administration is via oxidative DNA damage and up-regulation of NO/TNF-α and caspase 3 activities. PLoS ONE, 15(3): e0224052.
Lina S, Eliza H, Hashida NH, Ibrahim SF and Osman K. (2018). Androgen receptor and ultrastructural features of Nigella sativa oil and nicotine-treated male rat reproductive glands. Sains Malays, 47(8): 1827-1833.
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.


