Active Blood Pressure Lowering Fractions from the Aqueous Extract of the Leaves of Phyllanthus amarus Schum and Thonn (Euphorbiaceae)
https://doi.org/10.51412/psnnjp.2022.23
Keywords:
hypertensive, normotensive, fractionation, aqueous fraction, Phyllanthus amarusAbstract
Background: The blood pressure-lowering effffect of aqueous leaves extract of Phyllanthus amarus has been reported in earlier work in normotensive albino rabbits. The effffects of organic solvent fractions were evaluated in Wistar rats.
Methods: The aqueous fraction obtained from the crude extract of the leaves was evaluated for blood pressure-lowering effffect in anaesthetized normotensive Wistar rats, at the graded doses of 2.5-40 mg/kg after which it was subjected to vacuum liquid chromatography (VLC) using combinations of chloroform, ethyl acetate, methanol and water. This yielded eleven fractions which were bulked into six samples (A-F) after thin layer chromatograph analysis, and samples A, B and C were evaluated for activities on the blood pressure of hypertensive Wistar rats at the graded doses of 5 to 20 mg/kg. Fraction “C” showed the highest blood pressure lowering effffect and was further subjected to column chromatographic and VLC analysis, to obtain fraction (BVLC2) which was evaluated for possible blood pressure lowering effffect in hypertensive Wistar rats.
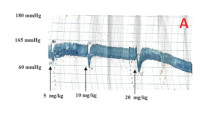
Results: The aqueous fraction caused a dose de pendent decrease in the blood pressure of normotensive Wistar rats, decreasing the mean arterial blood pressure (MAP) from a basal level of 104.58 ± 8.29 mmHg to 45.22 ± 6.71 mmHg (p<0.0001; 66.4%) at the dose of 40 mg/kg. Fraction “C” caused signifificant decrease in the MAPfrom 116.99 ± 10.28 mmHg to 68.33 ± 6.78 mmHg (p < 0.001; 0.01%) at the dose of 20 mg/kg, while the fraction BVLC2 decreased the MAPfrom 146.11 ± 8.29 mmHg to 79.33 ± 6.18 mmHg (***p< 0.001) at the dose of 10 mg/kg, indicating a trend of increasing potency along the course of purifification.
Conclusion: The aqueous fraction of the leaves of Phyllanthus amarus yielded column chromatographic sample (BVLC2), that might possibly serve as a lead sample from which pure active hypotensive constituent(s) could be isolated.
References
Etuk EU (2006). A Review of Medicinal Plants with Hypotensive or Antihypertensive effects. Journal of Medical Sciences 6: 894 - 900. http://doi.org/10.3923/jms.2006.894.900
Tamargo J, Duarte J, Ruilope LM (2015). New antihypertensive under development. Current Medicinal Chemistry. Volume 22; (3); 305 - 347. http://doi.org/10.2174/0929867321666141106113018
AlDisi SS, Anwar MA, Eid AH (2016). Antihypertensive Herbs and Their Mechanisms of Action. Frontiers in Pharmacology_ Cardiovascular and Smooth Muscle. hƩp://doi.org/10.3389/fphar.2016.00050
Susalit E, Agus N, Effendi I, Tjandrawinata RR, Nofiarny D, Perrinjaquet-Moccetti T (2011). Olive (Olea europaea) leaf extract effective in patients with stage-1 hypertension: comparison with Captopril. Phytomedicine. 18, 251 – 258. http://doi.org/10.1016/j.phymed.2010.08.016
Wang HP, Yang J, Qin LQ, Yang XJ (2015). Effect of garlic on blood pressure: a meta-analysis. J. Clin. Hypertens. 17, 223 – 231.
http://doi.org/10.1111/jch.12473
Tabassum N, Ahmad F (2011). Role of Natural Herbs in the Treatment of Hypertension. Pharmacogn Rev. 5(9): 30-40. http://doi.org/10.4103/0973-7847.79097
August P (2004). Overview: mechanisms of hypertension: cells, hormones, and the kidney. J. Am. Soc. Nephrol. 15, 1971 – 1973 . http://doi.org/10.1097/01.ASN.0000133197.23478.76
Wadood A, Ghufran M, Jamal SB, Naeem M, Khan A, Ghaffar R and Asnad (2013). Phytochemical Analysis of Medicinal Plants Occurring in Local Area of Mardan. Biochemistry and Analytical Biochemistry. 2: 144. hƩp://doi.org/10.4172/2161-1009.1000144
Tariq AL and Reyaz AL (2013). Significance and Importance of Phytochemical present in Terminalia chebula. International Journal of Drug Development and Research. Vol. 5; Issue 3. ISSN 0975-9344
Ama e c h i n a FC Omo g b a i EKI ( 2 0 0 7 ). Hypotensive effect of the aqueous extract of the leaves of Phyllanthus amarus Schum and Thonn, (Euphorbiaceae). Acta Poloniae Pharmaceutica-Drug Research. 64: 547-52. ISSN 0001-6837
National Institute of Health, USA. Public health service policy on humane care and use of laboratory animals; 2002.
Badyal DK, Lata H, Dadhuchi AP(2003). Animal Models of Hypertension. Indian Journal of Pharmacology. 35: 349 – 362.
https://www.researchgate.net.>post
Krishnananda PI, Amit GD, Dipika AP, Mahendra SD, Mangesh PM, Vaibkhar CK (2017). Phytochemicals: Extraction methods, identification and detection of bioactive compounds from plant extracts. Journal of Pharmacognosy and Phytochemistry. 6 (1): 32-36. ISSN 2278-4136
Cos P, Vlientinck AJ, Berghe DV, Maes L (2006). Anti-infective potentials of natural products: How to develop a stronger in vitro proof – of – concept. Journal of Ethnopharmacology, 106; 290 - 302. http://doi.org/10.1016/j.jep.2006.04.003
Mosic M, Dramicanin A (2020). Extraction as a Critical Step in Phytochemical Analysis. Journal of AOAC International, 103 (2) : 365 - 372. http://doi.org/10.5740/jaoacint.19-0251
Demers D, Wachs D (2021). Physiology, Mean Arterial Pressure. StatPearls Publishing NCBI Bookshelf.htm#_NBK538226_ai .s
Views | PDF Downloads:
595
/ 313
/ 0
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.


