Preliminary study on the anti-bacterial activity of 2 cultivars of Acalypha wilkesiana on bacterial isolates of clinical significance
https://doi.org/10.51412/psnnjp.2022.20
Keywords:
Agar diffffusion, Acalypha wilkesiana, Anti-bacterial, In-vitroAbstract
Background: Anti-microbial resistance has become a major problem in clinical practice making it necessary to search for newer molecules. Acalypha wilkesiana, is a herb commonly used traditionally in Nigeria to treat gastrointestinal disorders, wounds and skin infections like impetigo.
Objectives: To determine the in-vitro anti-bacterial activity of crude extracts of 2 cultivars of Acalypha wilkesiana (Macrophylla & Hoffffmanii)
Methods: In-vitro antibacterial activity was investigated by agar diffffusion and micro-broth dilution techniques. Bacterial isolates tested were Escherichia coli, Methicillin Resistant Staphylococcus aureus, Staphylococcus aureus, Salmonella paratyphi, Pseudomonas aeruginosa and Klebsiella pneumoniae.
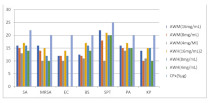
Results: The ethanol extract of both cultivars were more active than the ethyl acetate and n-hexane extracts having diameter zones of inhibition against all the bacteria tested at a range of 12.00-22.00 The lowest MIC of the crude ethanol extract was seen with E. coli (1000 µg/ml) and S. aureus (1000 µg/ml; 2000 µg/ml) while the most active fractions (R2, R4, G3 and G4) of the crude ethanol extract of both cultivars had greatest activity against MRSAand P. aeruginosa (156 µg/ml).
Conclusion: This study shows that Acalypha wilkesiana has good broad spectrum anti-bacterial activity with the potential for development as lead compounds to combat multi-drug resistant organisms like MRSA.
References
Organization WH. The evolving threat of antimicrobial resistance: options for action. 2012. Geneva, Switzerland: World Health Organization. 2013
Levy SB. (2000): Antibiotic and antiseptic resistance: impact on public health. The Pediatric Infectious Disease Journal. 19(10): S120-S122
Powe rs J. (2004): Antimi c robi a l drug development–the past, the present, and the future. Clinical Microbiology and Infection. 10:23-31.
Organization WH. Antimicrobial resistance global report on surveillance: 2014 summary. World Health Organization, 2014.
Thabit AK, Crandon JL, Nicolau DP. (2015): Antimicrobial resistance: impact on clinical and economic outcomes and the need for new antimicrobials. Expert Opinion on Pharmacotherapy. 16 (2): 159 - 177. https//doi.org/10.1517/146566.2015.993381
Grundmann H, Aires-de-Sousa M, Boyce J, Tiemersma E. (2006): Emergence and resurgence of meticillin-resistant Staphylococcus aureus as a public-health threat. The Lancet. 368(9538): 874-885. https//doi.org/10.1016/S0140-6736(06)68853-3
Kapil A. (2005): The challenge of antibiotic resistance: need to contemplate. Indian Journal of Medical Research. 121(2):83-91. PMID: 15756040
ECDC E. (2009): The bacterial challenge: time to react. Stockholm: European Center for Disease Prevention and Control.
Bryskier A. Antimicrobial agents: Antibacterials and antifungals: ASM press; 2005.
Efferth TE. From ancient herb to modern drug: Artemisia annua and artemisinin for cancer therapy. Seminars in cancer biology; 2017: Elsevier.
Organization WH. (2002): Traditional medicinegrowing needs and potential. WHO policy perspectives on medicine, No. 2. WHO/EBM/2002. WHO: Geneva.
Gupta PD, Daswani PG, Birdi TJ. (2014): Approaches in fostering quality parameters for medicinal botanicals in the Indian context. Indian Journa l of Pha rma cology. 246(4):363. https//doi.org/10.4103/0253-7613.135946
Lewis K, Ausubel FM. (2006): Prospects for plant-derived anti-bacterial. Nature Biotechnology. 24(12): 1504.
https//doi.org/10.1038/nbt1206-1504
Ikewuchi C, Ikewuchi C, Onyeike E, Uwakwe A. (2010): Nutritional potential of the leaves of Acalypha wilkesiana Godseffiana'Muell Arg. Journal of Applied Sciences and Environmental Management. 14(3): 21 - 24.
https//doi.org/10.4314/jasem.v14i3.61454
Iniaghe OM, Malomo SO, Adebayo JO. (2009): Proximate composition and phytochemical constituents of leaves of some Acalypha species. Pakistan Journal of Nutrition. 8(3):256-258. https//doi.org/10.3923/pjn.2009.256.258
Emeka PM, Badger-Emeka LI, Fateru F. (2012): In-vitro antimicrobial activities of Acalypha ornate leaf extracts on bacterial and fungal clinical isolates. Journal of Herbal Medicine. 2(4):136-142. https//doi.org/10.1016/J.HERMED.2012.09.001
Ikewuchi JC, Onyeike EN, Uwakwe AA, Ikewuchi CC. (2011): Effect of aqueous extract of the leaves of Acalypha wilkesiana Godseffiana Muell Arg (Euphorbiaceae) on the hematology, plasma biochemistry and ocular indices of oxidative stress in alloxan induced diabetic rats. Journal of Ethnopharmacology. 137(3):1415-1424. https//doi.org/10.1016/j.jep.2011.08.015.
Iniaghe OM, Egharevba O, Oyewo EB. (2013): Effect of aqueous leaf extract of Acalypha wilkesiana on hematological parameters in male Wistar albino rats. British Journal of Pharmaceutical Research. 3(3):465-71. https//doi.org/10.9734/BJPR/2013/2601
Awe F, Giwa-Ajeniya A, Akinyemi A, Ezeri G. (2013): Phytochemical analysis of Acalypha wilkesiana, Leucaena leucocephala, Pepperomia pellucida and Senna alata leaves. The International Journal of Engineering and Sciences (IJES). 2(9):41-44.
Ogundaini A. From greens into medicine: Taking a lead from nature. 2005. Inaugural lecture. Obafemi Awolowo University, Ile Ife, Nigeria
Sharma B, Kumar P, Joshi SC. (2011): Topical treatment of dermatophytic lesion on mice (Mus musculus) model. Indian Journal of Microbiology. 51(2):217–222. https//doi.org/10.1007/s12088-011-0109-2
Masoko P, Eloff JN. (2005): The diversity of antifungal compounds of six South African Terminalia species (Combretaceae) determined by bioautography. African Journal of Biotechnology. 4(12): 1425 -1431.
Pooja A, Arun N and Maninder K. (2013): Screening of plant essential oils for antifungal activity against Malassezia furfur. International Journal of Pharmaceutical Science. 2014;5(2): 37-39.
Santhanam J, AbdGhani FN, Basri DF. (2014): Antifungal activity of Jasminum sambac against Malassezia sp. and non-malassezia sp. isolated from human skin samples. Journal of Mycology. 2014: 1-7. https//doi.org/10.1155/2014/359630.
Othman M, Genapathy S, Liew PS, Ch'ng QT, Loh HS, Khoo TJ, et al. (2011): Search for antibacterial agents from Malaysian rainforest and
tropical plants. Natural Product Research. 25(19):1857-1864. https//doi.org/10.1080/14786419.2010537274.
Alade P, Irobi O. (1993): Antimicrobial activities of crude leaf extracts of Acalypha wilkesiana. Journal of Ethnopharmacology. 39(3):171-174. https//doi.org/10.1016/0378-8741(93)90033-2
Akinyemi KO, Oladapo O, Okwara CE, Ibe CC, Fasure KA. (2005): Screening of crude extracts of six medicinal plants used in South-West Nigerian unorthodox medicine for anti-methicillin resistant Staphylococcus aureus activity. BMC Complementary Medicine and therapies. 5(1):6. https//doi.org/10.1186/1472-6882-5-6.
Adetutu A, Morgan WA, Corcoran O. (2011): Ethnopharmacological survey and in vitro evaluation of wound-healing plants used in Southwestern Nigeria. Journal of Ethnopharmacology. 137 (1) : 50 - 56. https//doi.org/10.1016/j.jep.2011.03.073.
Parekh J, Jadeja D and Chanda S. (2005): Efficacy of aqueous and methanol extracts of some medicinal plants for potential antibacterial activity. Turkish Journal of Biology. 29:203-210. https//doi.org/10.1.1.452.9330.
Adhika P, Pandey A, Agnihotri V, Pande V, Van Vuuren SF. (2008): Antimicrobial activity of South African medicinal plants. Journal of Ethnopharmacology. 119 : 462 - 472. https//doi.org/10.1016/j.jep.2008.05.038.
Parekh J, Karathia N, Chanda S. (2006): Screening of some traditionally used medicinal plants for potential antibacterial activity. Indian Journal of Pharmaceutical Sciences. 68: 832-834. https//doi.org/10.4103/0250-474X.31031.
Oladunmoye, M. K. (2010): Effect of concentration on the rate of killing of some microorganisms and haemolytic activity of two varieties of Acalypha wilkesiana. Research Journal of Microbiology. 5(11): 1175-1179.
Fadeyi A, Bolaji B, Oyedepo O, Adesiyun O, Adeboye M, Olanrewaju T, Aderibigbe A, Salami A, Desalu O, Fowotade A, Nwabuisi C, Akanbi A, Raheem R, Olalere A. (2010): Methicilin Resistant Staphylococcus aureus Carriage amongst Healthcare Workers of the Critical Care Units in a Nigerian Hospital. American Journal of Infectious Diseases. 6(1), 18 - 23.
https://doi.org/10.3844/ajidsp.2010.18.23
Santiago C, Ee Leen P, Kuan-Hon L, Hwei-San L and Kang NT. (2014): Reversal of ampicillin resistance in MRSA via inhibition of penicillinbinding protein 2a by Acalypha wilkesiana. Biomed Research International. 2014:1-7. https//doi.org/10.1155/2014/965348
Thabit AK, Hobbs ALV, Guzman OE, Shea KM. (2019): The Pharmacodynamics of Prolonged Infusion β-Lactams for the Treatment of Pseudomonas aeruginosa Infections: A systematic review. Clinical Therapeutics. 41(11): 2397 - 2415. e8.doi:
1016/j.clinthera.2019.09.010. Epub 2019 Nov 1. PMID: 31679822.
Adesina SK, Idowu O, Ogundaini AO, Oladimeji H, Olugbade TA, Onawunmi, GO and Pais M. (2000): Antimicrobial constituents of the leaves of Acalypha wilkesiana and Acalypha hispida. Phytotherapy Research. 14:371-374
Awe, S and Eme I. (2014): Antibacterial activity and phytochemical screening of Acalypha wilkesiana (Copper Leaf) leaf extract on some clinical isolates. Res J Pharm Bio Chem Sci. 5(4), 1-7
Oladunmoye MK. (2006): Comparative Evaluation of Antimicrobial Activities and Phytochemical Screening of Two Varieties of
Acalypha wilkesiana. Trends in Applied Scientific Research. (1): 538–541.
Ubani-Ukoma UN, Ilomuanya MO, Amos SI, Aboh M, Katibi OS, Azubuike CP, Salawu OA. (2021): Evaluation of the Antimicrobial Effectiveness of Topical Gels Containing Acalypha wilkesiana. Tropical Journal of Natural Product Research. 5(5) : 1470 - 1477. http://www.doi.org/10.26538/tjnpr/v5i8.24
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.


