Vegetative anatomical characteristics and trichome morphology of the monotypic Martynia annua L. (Martyniaceae) for use in identification and crude drug search
https://doi.org/10.51412/psnnjp.2022.27
Keywords:
vegetative and reproductive parts, taxonomy, pharmacognosy, light microscopy, EudicotAbstract
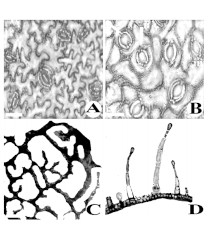
Background: Leaf epidermis and indumenta structure of M. annua were studied with the aid of microscopy for easy identification of the plant materials when in fragments and for crude drug search. Stomata and trichome morphology appear significant in identifying the species. Paracytic and anomocytic stomatal types are basic but polocytic is additional on the abaxial surface of the
epidermis.The overwhelming reports on the folkloric uses of Martynia annua L. (Martyniaceae) and the sole reliance on exo-morphology for its identification which gives room to error form the basis for this study. Moreso, the need to utilize as many data as possible for accurate identification of the plant is another important reason for its choice for investigation.
Methods: A total of 20 plant samples were examined. 100 leaves and 40 stems, petioles and midribs obtained from both herbarium samples of the plant were examined. The epidermis was recovered after treatment with concentrated trioxonitrate (v) acid (HNO3) in capped specimen bottles for about 8–24hrs to macerate the mesophyll. Samples were stained with acidified phloroglucinol and then one drop of toluidine blue. They were mounted on glass slides and examined under the microscope (XSZ- 170BN Olympus) and photomicrographs were taken with microscope eyepiece camera (Toupview 3.7). Line drawings were made with camera lucida drawing instrument.
Results: Interesting folia epidermal features were encountered. These include 3 stomatal types, two main types of trichomes namely: - Type 1: Long to short multicellular-glandular trichomes with globular heads and Type 2: Unicellular non-glandular globular-headed trichomes with stalk or no stalk.
Conclusion: The report of leaf teeth in M. annua which is the begonioid type is novel and cell shape of the perivascular structures is oval to polyhedral.Asuite of these characters will aid identification of M. annua when a whole fresh plant specimen is absent.
References
Richard H 1969. Nomenclatural History and Typification of Martynia and Proboscidea (Martyniaceae). Taxon, 18 (5): 527-534.
Willis JC 1973. A Dictionary of the flowering plants and ferns. Cambridge University Press, Cambridge. 1213pp.
Kenwat R, Prasad P, Satapathy T, Roy A 2013. Martynia annua: An Overview. UK Journal of Pharmaceutical and Biosciences, 1(1): 7-10.
Hutchinson J, Dalziel JM 1958. Flora of West Tropical Africa. White Friars Press Ltd, London.295pp.
Inamdhar JA, Gangadhara M 1977. Studies on the trichomes of some Euphorbiaceae. Feddes Repertorium, 88: 1033–111.
Davis PH, Heywood VH 1963. Principles of Angiosperm Taxonomy. Oliver and Boyd. Ltd., Edinburgh. 556 pp.
Santram L, Singhai AK 2011. Preliminary pharmacological evaluation of Martyni annua Linn leaves for wound healing. Asian Pacific
Journal of Tropical Biomedicine, 1(6): 421–427.8. Malik JK, Katare V 2020. Pharmacological activities of Martynia (Martynia annua Linn.): A
Brief Review. Asian Plant Research Journal, 4(2): 46-51.
Akin-Obasola BJ, Obe BW, Adewumi AE 2021. Effect of Martynia nnua (Devil's Claw) Leaf meal on the reproductive and growth performance of Oreochromis niloticus (Nile tilapia). Pakistan Journal of Nutrition, 20: 18-24.
Ajayi GO, Kadiri AB, Ibiloye AE 2015. Pharmacognostic characteristics of the West African Jateorhiza macrantha (Hook.F.) Exell &
Mendonça (Menispermaceae) International Journal of Pharmacognosy and Phytochemical Research, 7(5): 1001-1006. 11. Kadiri AB, Ayodele AE 2010. Anatomical characteristics of some commercial timbers from Nigeria. 1. Structures of wood elements. Nigerian
Journal of Botany, 23(1): 143-150.
Wilkinson HP 1989. Leaf Anatomy of Menispermaceae tribe Tilicoreae Miers. Botanical Journal of the Linnean Society, 125-174.
Kadiri AB, Olowokudejo JD 2016. Systematicsignificance of foliar epidermis and tendril morphology in three West African genera of Cucurbitaceae: Momordica L., Luffa Mill. and Trichosanthes L, Webbia, 71(1): 91-105.
Ogundipe OT, Kadiri AB 2012. Comparative foliar epidermal morphology of the West African species of Amaranthaceae Juss. Feddes
Repertorium, 123: 97–116.
, Carson FL 1997. Histotechnology. A SelfInstructional Text. American Society of Clinical Pathologists, Chicago. 160pp.
Cutler DF 1978. Applied Plant Anatomy. Longman Group Limited. NewYork. 103pp
Metcalfe CR, Chalk L 1950. Anatomy of Dicotyledons. Clarendon Press, Oxford. 724pp.
Metcalfe CR, Chalk L 1979. Systematic Anatomy of the leaf and stem. In: Anatomy of Dicotyledons. Oxford Science Publication. Great Britain. 276pp
Stace CA1965. Cuticular studies as an aid to plant taxonomy. Bulletin of the British Museum (Natural History) Botany 4: 1–71.
Radford AE, Dickson WC, Massey JR, Bell CR 1976. Vascular Plant Systematics. Harper and Row, USA. 886pp.
Kadiri AB, Zeng G, Liu B, Ferguson DK, Yang Y 2021. Leaf epidermal micromorphology of Beilschmiedia Nees (Lauraceae) from Africa.
T a i w a n i a , 6 6 ( 3 ) : 3 7 8 ‒ 3 8 . D O I : 10.6165/tai.2021.66.378.
Kadiri A B, Muellner-Riehl A N 2021 . Comparative leaf micromorphology of Drypetesand Putranjiva (Putranjivaceae) and its taxonomic significance. Botanical Journal of the Linnean Society, 195(2): 139-160.
Akwu NA, Naidoo Y, Singh M 2020. The anatomy and histochemistry of Grewia lasiocarpa E. Mey. ex Harv. (Malvaceae). SuidAfrikaanse
Tydskrif vir Natuurwetenskap en Tegnologie, 39(1): 1-17.
Geoffrey M 1995. Seed trichome variation in Hibiscus section Furcaria in East Africa Journal of East African Natural History, 84 (1): 65-75.
Ančev M, Goranova V 2006. Trichome morphology of eleven genera of the Tribe Alysseae (Brassicaceae) occurring in Bulgaria.Willdenowia 36: 193–204.
Zhang X, Lyu J, ZengY, Sun N, Liu C,Yin S 2021. Individual effects of trichomes and leaf morphology on PM2.5 dry deposition velocity: A
variable-control approach using species from the same family or genus. Environmental Pollution, 272: 1-8.
de Almeida-Jr. EB, Araújo JS, Santos-Filho FS, Zickel CS. 2012. Leaf morphology and anatomy of Manilkara Adans. (Sapotaceae) from
northeastern Brazil. Plant Systematics and Evolution, 299: 1–9.
Okeke CU, Iroka CF, Izundu AI, Okeke NC, Onwuasoeje C I , Nyananyo BL 2015 . Comparative systematic leaf and petiole
anatomical studies of the genus Stachytarpheta found in Awka, Nigeria. Journal of Medicinal Plant Studies, 3(4): 82-84.
Anorue GU, Olowokudejo JD, Kadiri AB 2020. Petiole anatomy of some selected species of the family Verbenaceae (Lamiales) in Nigeria. Indian Journal of Plant Sciences, 9: 1-14.
Riosi ABM, Menino GCO, Dalvi VC 202. Leaf teeth in eudicots: what can anatomy elucidate? Botanical Journal of the Linnean Society, 193: 504–522.
Gonzalez AM, Tarragó JR 2009. Anatomical structure and secretion compounds of colleters in nine Ilex species (Aquifoliaceae) from southern South America. Botanical Journal of the Linnean Society, 160: 197–210.
Thomas V, Dave Y 1989. Structure, origin, development and senescence of colleters in Nerium indicum Mill. (N. odorum Soland.,
Apocynaceae). Korean Journal of Botany, 32: 163–172.
Thomas V, Dave Y 1990. Structure and necrosis of stipular colleters in Mitragyna parvifolia (Rubiaceae). Belgian Journal of Botany, 123 67- 72.
Thomas V 1991. Structural, functional and phylogenetic aspects of the colleter. Annals of Botany, 68: 287–305.
Kadiri AB, Agboola OM, Fashina FO 2013. Ethnobotanical survey and phyto-anatomical studies of some common plants used for the
treatment of epilepsy in some rural areas of South west Nigeria. Journal of Pharmacognosy and Phytochemistry, 2(4):175-182.
Holmgren PK, Keuken W, Schofield EK 1990. Index Herbariorum, Part 1: The herbaria of theWorld. Regnum Vegetabilis, 106: 1–456.
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.


