Musa acuminata Colla Millsp ameliorates paraquat-induced memory deficit and oxidative stress in murine Parkinson disease model
Keywords:
Catalepsy, Musa acuminata, paraquat, Parkinsonism, oxidative stress, rotarod testAbstract
Background: Parkinson disease (PD) is one of the most common neurodegenerative disorders and remains an unmet medical need. A number of epidemiological as well as case-control studies have revealed an association between pesticide exposure, especially that of paraquat (PQ) and occurrence of Musa acuminata Colla (Musaceae) also known as banana is widely cultivated for its delicious fruits and is one of the most economically important crops in the world. This study was designed to investigate the protective effffect of M. acuminata fruit extract (MAC) on paraquat-induced Parkinsonism in mice.
Methods: Sixty male albino mice were randomly divided into 6 groups (n=10) and treated as follows for 21 consecutive days; Group 1: vehicle control (10 ml/kg, p.o.), group 2: vehicle + paraquat 10 mg/kg, i.p., group 3: MAC (400 mg/kg, p.o), groups 4-6: MAC (100, 200 or 400 mg/kg, p.o.) + paraquat, respectively. Behavioural studies (rotarod, Y-maze, open fifield test and bar test) were carried out on weekly basis. On day 21, one hour post-treatment the midbrains were isolated for biochemical estimation of oxidative stress parameters.
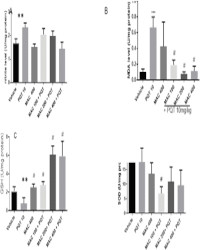
Results: MAC (100, 200 or 400 mg/kg) failed to reverse paraquat induced signifificant decrease in latency to fall in rotarod test. Similarly, MAC did not affffect paraquat-induced decrease in locomotor activity in open fifield test. MAC prevented paraquat-induced decrease in percent alternation behaviour in Y-maze task. Conversely, MAC aggravated paraquat-induced cataleptic behaviour in bar test. The pretreatment of mice with MAC (200 or 400 mg/kg) signifificantly reversed paraquat-induced oxidative stress parameters in the midbrain.
Conclusion: Findings from this study showed that M. acuminata prevented paraquat-induced memory impairment through improvement in antioxidant defense mechanism but failed to reverse paraquat induced locomotor defificit and cataleptic behaviour.
References
Samii A, Nutt JG, Ransom BR. (2004). Parkinson's disease. Lancet.;363(9423):1783-93. https://doi.org/10.1016/S0140-6736(04)16305-8
Ascherio A, Schwarzschild MA. (2016). The epidemiology of Parkinson's disease: risk factors and prevention. Lancet Neurology;15(12):1257-1272. https://doi:10.1016/S1474-4422(16)30230-7
Opara J, Małecki A, Małecka E, Socha T. (2017). Motor assessment in Parkinson`s disease. Annals of Agricultural and Environmental Medicine; 24(3): 411 - 415. https://doi.org/10.5604/12321966.1232774
Hayes MT. (2019). Parkinson's Disease and Parkinsonism. American Journal of Medicine. 132(7): 802 - 807. https://doi.org/10.1016/j.amjmed.2019.03.001
Breckenridge CB, Berry C, Chang ET, Sielken RL Jr, Mandel JS. (2016). Association between Parkinson's Disease and Cigarette Smoking, Rural Living, Well-Water Consumption, Farming and Pesticide Use: Systematic Review and Meta- Ana lysis. PLoS One . 11(4): e0151841. https://doi.org/10.1371/journal.pone.0151841
Chinta SJ, Woods G, Demaria M, Rane A, Zou Y, McQuade A, Rajagopalan S, Limbad C, Madden DT, Campisi J, Andersen JK. (2018). Cellular Senescence Is Induced by the Environmental Neurotoxin Paraquat and Contributes to Neuropathology Linked to Parkinson's Disease. Cell Reports. (4): 930 - 940. https://doi.org/10.1016/j.celrep.2017.12.092
Blandini F, Armentero MT. (2012). Animal models of Parkinson's disease. FEBS Journal. ;279(7):1156-66. https://doi.org/10.1111/j.1742-4658.2012.08491.x
Mathew NS, Negi PS. (2017). Traditional uses, phytochemistry and pharmacology of wild banana (Musa acuminata Colla): A review. Journal of Ethnopharmacology, 196: 124 - 140. https://doi.org/10.1016/j.jep.2016.12.009
Swanson MD, Winter HC, Goldstein IJ, Markovitz DM. (2010). A lectin isolated from bananas is a potent inhibitor of HIV replication. Journal of Biological Chemistry; 285(12):8646-55. https://doi.org/10.1074/jbc.M109.034926
Vijayakumar K, Ramanathan T. (2020). Musa acuminata and its bioactive metabolite 5-Hydroxymethylfurfural mitigates quorum sensing (las and rhl) mediated biofilm and virulence production of nosocomial pathogen Pseudomonas aeruginosainvitro. Journal of Ethnopharmacology; 246: 112242. https://doi.org/10.1016/j.jep.2019.112242
Oresanya IO, Sonibare MA, Gueye B, Balogun FO, Adebayo S, Ashafa AOT, Morlock G. (2020). Isolation of flavonoids from Musa acuminata Colla (Simili radjah, ABB) and the in vitro inhibitory effects of its leaf and fruit fractions on free radicals, acetylcholinesterase, 15- lipoxygenase, and carbohydrate hydrolyzing enzymes. Journal of Food Biochemistry;
(3):e13137. https://doi.org/10.1111/jfbc.13137
Pothavorn P, Kitdamrongsont K, Swangpol S, Wongniam S, Atawongsa K, Savasti J, Somana J. (2010). Sap phytochemical compositions of some bananas in Thailand. Journal of Agricultural Food Chemistry, 58(15): 8 7 8 2 - 7 .
https://doi.org/10.1021/jf101220k PMID:20681667.
Ishola IO, Akataobi OE, Alade AA, Adeyemi OO. (2019). Glimepiride prevents paraquat-induced Parkinsonism in mice: involvement of oxidative stress and neuroinflammation. Fundamental and Clinical Pharmacology; 33(3):277-285. https://doi.org/10.1111/fcp.12434
Ishola IO, Katola FO, Adeyemi OO. (2020). Involvement of GABAergic and nitrergic systems in the anxiolytic and hypnotic effects of Curcuma longa: its interaction with anxiolytic-hypnotics. Drug Metabolism and Personalized Therapy.
https://doi.org/10.1515/dmdi-2020-0147
Adedeji HA, Ishola IO, Adeyemi OO. (2014). Novel action of metformin in the prevention of haloperidol-induced catalepsy in mice: Potential in the treatment of Parkinson's disease? Progress in Neuropsychopharmacology and Biological Psychiatry, 48: 245 - 251. https://doi.org/10.1016/j.pnpbp.2013.10.014
Ishola IO, Akinyede AA, Adeluwa TP, Micah C. (2018). Novel action of vinpocetine in the prevention of paraquat-induced parkinsonism in mice: involvement of oxidative stress and neuroinflammation. Metabolic Brain Disease;
(5): 1493 - 1500. https://doi.org/10.1007/s11011-018-0256-9
Ishola IO, Tota S, Adeyemi OO, Agbaje EO, Narender T, Shukla R. (2013). Protective effect of Cnestis ferruginea and its active constituent on scopolamine-induced memory impairment in mice: a behavioral and biochemical study. Pharmaceutical Biology, 51(7):825-35. https://doi.org/10.3109/13880209.2013.767360
Ishola IO, Jacinta AA, Adeyemi OO. (2019). Cortico-hippocampal memory enhancing activity of hesperetin on scopolamine-induced amnesia in mice: role of antioxidant defense system, cholinergic neurotransmission and expression of BDNF. Metabolic Brain Disease, 34(4):979-989. https://doi.org/10.1007/s11011-019-00409-0
Ishola IO, Isaac AI, Adeyemi OO. (2020). Vinpocetine prevents haloperidol-induced cognitive and working memory deficits through attenuation of oxidative and nitrosative stress in mice. Nigeria Journal of Physiological Sciences, 35(2):203-208.
Brooks AI, Chadwick CA, Gelbard HA, CorySlechta DA, Federoff HJ. (1999). Paraquat elicited neurobehavioral syndrome caused by dopaminergic neuron loss. Brain Research., 823(1-2):1-10. https://doi.org/10.1016/s0006-
(98)01192-5
Wadenberg ML, Kapur S, Soliman A, Jones C, Vaccarino F. (2000). Dopamine D2 receptor occupancy predicts catalepsy and the suppression of conditioned avoidance response behavior in rats. Psychopharmacology (Berl), 150(4):422-9. https://doi.org/10.1007/s002130000466
Burbulla LF, Song P, Mazzulli JR, Zampese E, Wong YC, Jeon S, Santos DP, Blanz J, Obermaier CD, Strojny C, Savas JN, Kiskinis E, Zhuang X, Krüger R, Surmeier DJ, Krainc D. (2017). Dopamine oxidation mediates mitochondrial and
lysosomal dysfunction in Parkinson's disease. Science, 357 (6357): 1255 - 1261. https://doi.org/10.1126/science.aam9080
Tanner CM, Kamel F, Ross GW, Hoppin JA, Goldman SM, Korell M, Marras C, Bhudhikanok GS, Kasten M, Chade AR, Comyns K, Richards MB, Meng C, Priestley B, Fernandez HH, Cambi F, Umbach DM, Blair A, Sandler DP, Langston JW. (2011). Rotenone, paraquat, and Parkinson's disease. Environment and Health Perspectives, 119(6): 866 - 72. https://doi.org/10.1289/ehp.1002839
Published
How to Cite
Issue
Section
License

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.


